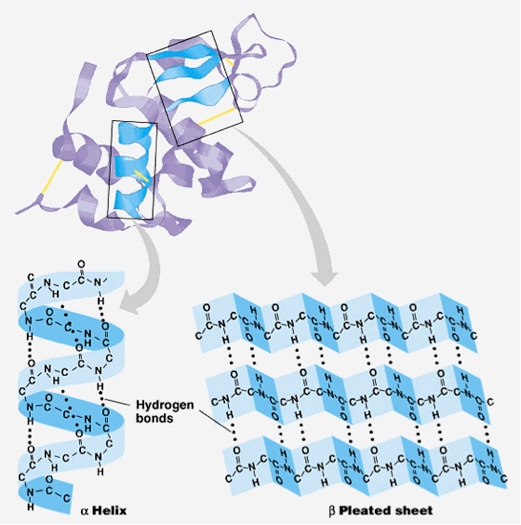
There are four distinct levels of protein structure. The alternative structures of the same protein are referred to as different conformations, and transitions between them are called conformational changes. For example, many thousands of actin molecules assemble into a microfilament.Ī protein usually undergoes reversible structural changes in performing its biological function. Very large protein complexes can be formed from protein subunits. By physical size, proteins are classified as nanoparticles, between 1–100 nm. 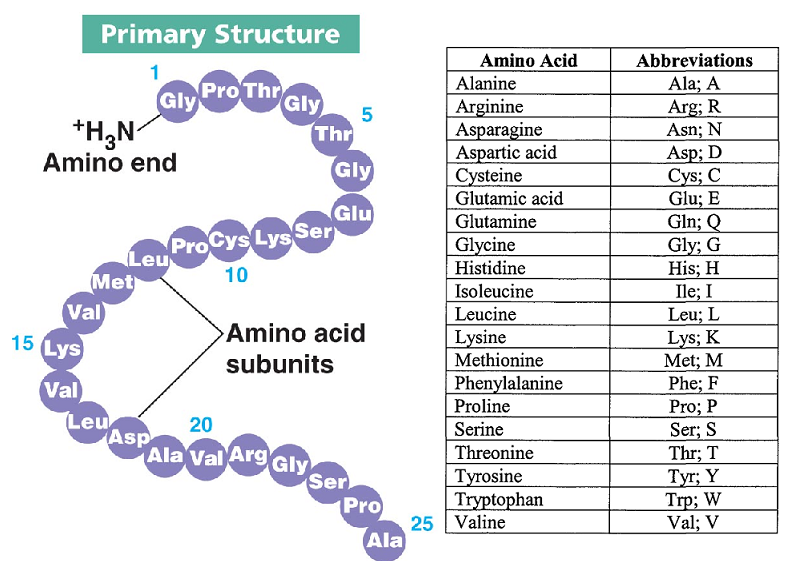
Protein structures range in size from tens to several thousand amino acids. This is the topic of the scientific field of structural biology, which employs techniques such as X-ray crystallography, NMR spectroscopy, cryo-electron microscopy (cryo-EM) and dual polarisation interferometry to determine the structure of proteins. To understand the functions of proteins at a molecular level, it is often necessary to determine their three-dimensional structure. To be able to perform their biological function, proteins fold into one or more specific spatial conformations driven by a number of non-covalent interactions such as hydrogen bonding, ionic interactions, Van der Waals forces, and hydrophobic packing. By convention, a chain under 30 amino acids is often identified as a peptide, rather than a protein. Proteins form by amino acids undergoing condensation reactions, in which the amino acids lose one water molecule per reaction in order to attach to one another with a peptide bond. A single amino acid monomer may also be called a residue indicating a repeating unit of a polymer. Proteins are polymers – specifically polypeptides – formed from sequences of amino acids, the monomers of the polymer. Protein structure is the three-dimensional arrangement of atoms in an amino acid-chain molecule. This diagram (which is interactive) of protein structure uses PCNA as an example.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
